 This beam was deflected toward the positive charge and away from the negative charge, and was produced in the same way with identical properties when different metals were used for the electrodes. When high voltage was applied across the electrodes, a visible beam called a cathode ray appeared between them. This apparatus consisted of a sealed glass tube from which almost all the air had been removed the tube contained two metal electrodes. If matter were composed of atoms, what were atoms composed of? Were they the smallest particles, or was there something smaller? In the late 1800s, a number of scientists interested in questions like these investigated the electrical discharges that could be produced in low-pressure gases, with the most significant discovery made by English physicist J. While the historical persons and dates behind these experiments can be quite interesting, it is most important to understand the concepts resulting from their work. Here, we will discuss some of those key developments, with an emphasis on application of the scientific method, as well as understanding how the experimental evidence was analyzed. Much of this came from the results of several seminal experiments that revealed the details of the internal structure of atoms. In the two centuries since Dalton developed his ideas, scientists have made significant progress in furthering our understanding of atomic theory. These concepts form the basis of chemistry. Atoms combine in whole-number ratios to form compounds.Atoms of the same element are the same atoms of different elements are different.The concept that atoms play a fundamental role in chemistry is formalized by the modern atomic theory, first stated by John Dalton, an English scientist, in 1808. Atoms are so small that it is difficult to believe that all matter is made from atoms-but it is. The period at the end of a printed sentence has several million atoms in it. It would take about fifty million atoms in a row to make a line that is 1 cm long. 
The smallest piece of an element that maintains the identity of that element is called an atom. Write isotopic symbols for elements and ions.Define isotopes and give examples for several elements.Describe the three subatomic particles that compose atoms. 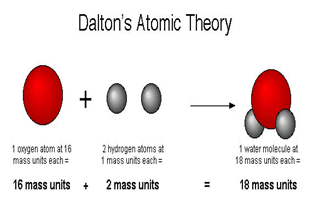 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
